The Secret Physics of Nuclear Weapons
The development of nuclear weapons has been one of the most pivotal events of the modern era. With the successful test of a 21 kiloton device in New Mexico in 1945, along with two wartime bombs deployed against Japan shortly thereafter, The United States became the first major power to unveil this frightening technology to the world.
Not wanting to be left behind, the other major powers nominally victorious in the Second World War, also sought to develop their own nuclear arsenals; the desire for this fantastic new weapon proving quite irresistible. But how indeed to achieve it?
Any orthodox historical analysis will confirm that two things are essential:
1) Industrial capacity. The means to mine and process the necessary raw materials. The development of nuclear reactors for plutonium production. For this, any aspiring power has to have a strong technological-industrial base.
2) Cracking the actual ‘secret of the bomb.’ Determining just how to unleash the energy of the atom – specifically of Uranium and Plutonium. How to practically engineer a functioning nuclear device.
Certainly it is the second category that holds the greater interest; being the focus going forwards.

Above: Hiroshima and Nagasaki mushroom clouds (Japan, 1945)
Nuclear Weapons & Radioactive Decay
In the early years of the 20th century it was well understood from the basic E = mc^2 equation, that any given measure of matter – even a very small measure – contained a massive amount of energy.
Also, that the heaviest elements of the periodic table, beginning with Uranium (i.e. the transuranic elements), were naturally engaged in a process of radioactive decay. Essentially, they were continuously ‘casting off’ their matter into space in the form of radioactive particles: alpha, beta and gamma ray photons. Moreover, that the speed of emission of these particles at the point of decay was quite considerable. The most recent experimental speed values are as follows:
- Alpha decay = ~9940 miles per second, or about 5% of the speed of light
- Beta decay = ~182556 miles per second, or about 98% of the speed of light
- Gamma decay = 186282 miles per second, i.e. the speed of light
A lump of Uranium thus sat innocuously upon a desk is not merely an inactive substance, although to the naked eye it might appear so. At the atomic level it is exceptionally active, quite literally dispersing its own material throughout the universe continuously in all directions via the decay process.
Now although the various radioactive particles are shed at high velocity (beta and gamma being the fastest), the actual amount of physical matter released at such speeds over time is exceptionally small. And thus so too is the energy. So much so that over just a short period e.g. a few years, the total energy output in practical terms from a transuranic element undergoing radioactive decay is quite inconsequential. Indeed, due to the extremely low ‘energy/time’ output, one would have to wait literally hundreds of millions or even billions of years to realise a substantial amount of energy.
From such an insight, the key to developing a functional nuclear device was quickly realised.
Some way had to be found to dramatically speed up the process of radioactive decay - or an equivalent process - in the transuranic elements i.e. Uranium and Plutonium, thereby triggering the sudden release of a massive amount of energy that would ordinarily only be realised gradually over the course of many millions or billions of years. The outward product of such an accelerated action is indeed what actually constitutes a nuclear explosion.
Engineering A Nuclear Explosion
Two primary techniques are used to unlock the energy of the atom to produce a massive nuclear blast: Fission and fusion. Depending upon the design of a given nuclear bomb, either may be employed to disrupt the atomic structure of the main elements placed within it.
As a general rule there are two main types of nuclear weapons: Atom bombs and Thermonuclear bombs. The former are restricted to the fission process to produce their blast, which may range anywhere from a single kiloton (TNT) yield up to some 500 kilotons. The fusion process however can command yields in the multi-megaton range (TNT), and is capable of producing vastly more powerful explosions. It is the thermonuclear devices which make use of this technique.
One may further note that the elements themselves (as used within a nuclear bomb) determine which technique is applied to them.
Fission is usually applied to the two main transuranic elements: Uranium (235) and Plutonium (239); being the process of actually splitting the atoms of these heavy elements. A nuclear explosion being the direct product of the immense energy suddenly released by this action.
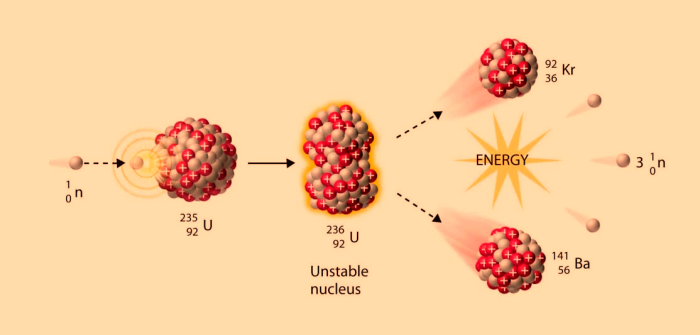
Above: Fission of Uranium-235: A uranium-235 atom absorbs a neutron to become a uranium-236 atom. Uranium-236 is unstable and fissions into two new atoms, releasing three new neutrons and some binding energy.
The fusion technique is the exact opposite of fission, involving fusing together i.e. combining elements. Specifically Deuterium and Tritium; being two isotopes of the hydrogen atom, the lightest element of the periodic table. This represents an alternate method of releasing a massive amount of energy.
Throughout the late 1940s only the fission process had yet been developed, and thus all of the nuclear devices tested during this time were atom bombs making use of Uranium or Plutonium. Although the theoretical basis of fusion was postulated during this period, it was only perfected and successfully tested in 1952. The critical test being conducted by the United States at Eniwetok Atoll, resulting in an extremely powerful thermonuclear blast with an explosive yield of some 10.4 Megatons of TNT (codenamed: Mike).
It is well to note that whereas atom bombs only incorporate fission in their design, thermonuclear devices incorporate both fission and fusion. Indeed, they are strictly speaking two stage devices. The fission process being triggered first, followed by the fusion reaction. To be sure, the energetic conditions first created in the device by the fission process are absolutely necessary to subsequently trigger the fusion process.
To date, all thermonuclear devices developed require an initial fission reaction to initiate fusion. As of this time no major power – at least officially – has been able to successfully develop a pure fusion based nuclear weapon.
Below: Castle-Bravo - USA test (1954): 15 Megatons Yield

The Main Sequence of a Nuclear Bomb
1) The Fission Process
A nuclear bomb is an extremely intricate device that relies on a complex series of stages to successfully function. Once triggered, the initial fission process begins when the transuranic core – usually Plutonium (239) – is bombarded by a stream of free neutron particles, causing the nuclei of the Plutonium atoms to split upon impact. Each instance resulting in the creation of two smaller more stable atoms, along with a simultaneous release of energy.
A Plutonium (239) atom thus fissioned may produce both Zirconium (103) and Xenon (134), plus some 3 additional neutrons [1], the latter of which may go on to strike other Plutonium atoms, splitting them also. If carefully sustained, the entire process becomes self amplifying. What is known to physics as a ‘chain reaction’ effect, without which a nuclear explosion is impossible to engineer. Achieving this is however a very delicate matter.
Under most conditions a standard Plutonium core subject to neutron bombardment is usually incapable of sustaining the fission process. Indeed, a significant measure of neutrons successively released by each ‘fission generation’ are invariably lost to the process; failing to go on to split new atoms in order to sustain it. To ensure that this does not happen, just prior to the neutrons being released, the Plutonium core is first ‘optimised’ to attain what is known as a supercritical state. This is accomplished by physically compressing it via an implosion process towards a metallic fluid-like state.
As is standard in almost all nuclear bomb designs, the Plutonium core is carefully surrounded on all sides by a series of explosive charges; all triggered in unison upon activation of the device. Upon sudden implosion to a high density supercritical state, the core is at the precise optimum, such that bombardment with neutrons at this point all but guarantees a successful chain reaction effect.
In order to actually produce the opening burst of neutrons to start the fission process, a special device known as a neutron initiator is used, making use of beryllium and polonium. In many weapons configurations the very centre of the Plutonium core is hollow; being the ideal location for the initiator. The explosive compression of the core directly activating it to produce the first neutron batch.
2) The Fusion Process
As previously noted, a thermonuclear bomb is a two stage device involving both fission and fusion; the former process being essential to initiating the latter.
The two key elements involved in fusion are deuterium and tritium. Both of which are isotopes of the basic hydrogen atom. The only difference is the presence of added neutrons at their core, giving them each a much heavier nucleus. One may compare all three as follows:
- Hydrogen = 1 proton with 1 electron.
- Deuterium = 1 proton and 1 neutron, with 1 electron.
- Tritium = 1 proton and 2 neutrons, with 1 electron.
In modern thermonuclear weapons the main fusion fuel is lithium-6 deuteride. A special mixture of deuterium, combined with the means to generate tritium. Indeed, tritium itself is not contained within the device itself in pure form. It is created as part of the overall fusion process. The key phase being when the lithium-6 deuteride mixture undergoes neutron bombardment.
With the extreme temperatures created from the initial fission stage of a nuclear bomb, the nuclei of the deuterium and tritium – successfully created in sequence – are fused together to form helium-4, including a high-energy neutron. The overall output being an exceptional release of energy.
Below: Modern Thermonuclear Warhead
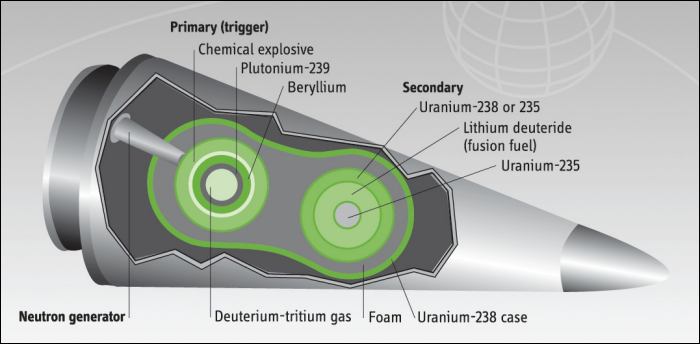
A modern thermonuclear warhead may contain both plutonium and highly enriched uranium (Average estimated values are 4 kg and 25 kg of plutonium and HEU, respectively).
Nuclear Fission / Fusion Efficiency
The efficiency of a nuclear bomb is typically expressed as a percentage of the energy released by the bomb against the total energy of the fuel. In other words, the percentage of the transuranic material within the device that is successfully fissioned, combined with that having undergone fusion (in the case of thermonuclear weapons).
Pure Fission Energy Conversion
In terms of a theoretical ideal, the complete fissioning of just 1 kg (2.2 pounds) of uranium-235 will produce an outward explosive yield of some 17000 tons (17 kilotons) of TNT. Of course in practical terms, a 100% ideal fissioning of any transuranic element is never achieved. Not even close. Indeed, in many cases most nuclear weapons fission only a fraction of the radioactive materials they contain. One may consider the two bombs dropped on Japan in 1945 by way of example:
Target: Hiroshima
- Explosive yield: 15000 Tons of TNT
- Gun type uranium-235 fission bomb
- Fuel: 64 kg of Uranium-235
- About 1.38% of the uranium fissioned
(The remainder, representing most of the entire wartime output of the giant Y-12 factories at Oak Ridge, scattered uselessly).
Target: Nagasaki
- Explosive yield: 20000 Tons of TNT
- Implosion type plutonium-239 fission bomb
- Fuel: 6.2 kg of Plutonium-239
- About 16% of the Plutonium fissioned (~1 kg)
Here then one can see that a considerable amount of Uranium was used in the Hiroshima bomb. A full 64 kilograms. And yet barely 1.4% underwent fission. In the case of the Nagasaki bomb, a far smaller amount of Plutonium was used. The efficiency here though being far higher, at some 16%.
Thermonuclear Considerations
Although thermonuclear weapons do have the added process of fusion, this secondary phase does not necessarily release more energy than the primary fission phase. In point of fact, fusion is usually employed as a means to produce an additional wave of fission reactions.
One may consider ‘Ivy Mike’ and ‘Castle Bravo’ as two prime examples of this. Both of these devices (US tests: 1952 & 1954 respectively) used the same weapons design, based upon the work of Stanislaw Ulam and Edward Teller. The Mike test, as previously noted, had a yield of some 10.4 megatons. Most of this came from fission however. An estimated 77% from the fast fission of the natural-uranium tamper of the device.
In the case of Castle Bravo at 15 megatons – the highest yield US test ever conducted – once again a substantial portion of the energy came from fission. In the design itself, the fission and fusion stages were kept physically separate in a reflective cavity. The radiation from the exploding fission primary brought the fuel in the fusion secondary to critical density and pressure, setting off thermonuclear (fusion) chain reactions, which in turn set off a tertiary fissioning of the bomb's Uranium 238 fusion tamper and casing. Consequently, this type of bomb is also known as a "fission-fusion-fission" device.
Overall, it was estimated that about 25% of the 880 pounds of lithium-6 deuteride used in the Bravo device underwent either fission or fusion.
Outward Nuclear Yield Determination
Calculating the yield of nuclear explosions, whether referring to estimates expressed in kilotons or megatons, is not without its difficulties. This being the case even under well controlled conditions, where the margins of error can still be very significant. In less controlled conditions, even more so.
With respect to pure fission devices, the most precise yield value can be obtained from a "radiochemical/Fallout analysis," involving the measurement of the quantity of fission products generated. A technique quite similar to the way in which a chemical yield from a chemical reaction may be determined. The radiochemical analysis method was pioneered by Herbert L. Anderson.
For nuclear explosive devices where the fallout is not attainable or would be misleading, neutron activation analysis is often employed as the second most accurate method, having been used to determine the yield of both Little Boy (Hiroshima, 1945) and Mike (Ivy test series, 1952).
If such data is not available, which is not uncommon, precise yield values may be greatly disputed. The weapons used to attack Hiroshima and Nagasaki for example were highly individual and very idiosyncratic designs. Gauging their yields retrospectively was thus difficult. Indeed, for the Hiroshima bomb the explosive yield was estimated at between 12 and 18 kilotons of TNT. The 15 kilotons value most often cited being nothing more than the average of the two, representing a 20% margin of error. In like manner, the estimated yield of the Nagasaki bomb is between 18 and 23 kilotons. Most references here settling upon 20 kilotons as an approximate average. This being in line with a 10% margin of error.
The Secret Missing Component
To realize the design yield of a nuclear bomb, it is essential to achieve a high level of efficiency in converting the primary plutonium fuel into energy. For all the testing that has been done over the years however, predicting the outward yield of a nuclear device purely from its design has never been achieved to anywhere near extreme accuracy. Persistent significant discrepancies have always remained, which cannot be merely waved away.
Now the reason for this – almost never discussed in conventional literature on nuclear weapons – is that the devices themselves are NOT CLOSED SYSTEMS. That is to say, that certain subtle natural energy fields that permeate space-time – being constantly in a state of flux – interact with the energetic functioning of a nuclear device upon activation. These ‘background energy fields,’ being continuously present, are an inescapable interference factor capable of significantly modifying the power (or efficiency) of the device.
Moreover, the interference of these external fields is directly linked to the major celestial bodies of the solar system. To be sure, as fantastic as it might seem, the positions of the very planets themselves, including the sun and moon, have a direct influence on the outward explosive yield of a nuclear device, when triggered.
To understand this point further one may view the following in-depth video I have produced:
A Secret Nuclear War is Happening Right Now
References
1) The average number of additional neutrons produced by Plutonium undergoing fission is about 2.89 from each neutron involved in splitting a Plutonium atom. NB; Zirconium (103) and Xenon (134) is just one possible combination of atoms produced from the fission of Plutonium. Perhaps the most common, but there are other combinations also.